Podcast: Play in new window | Download
This entire episode serves as cautionary advice to anyone seeking to add osmium to their element collection.
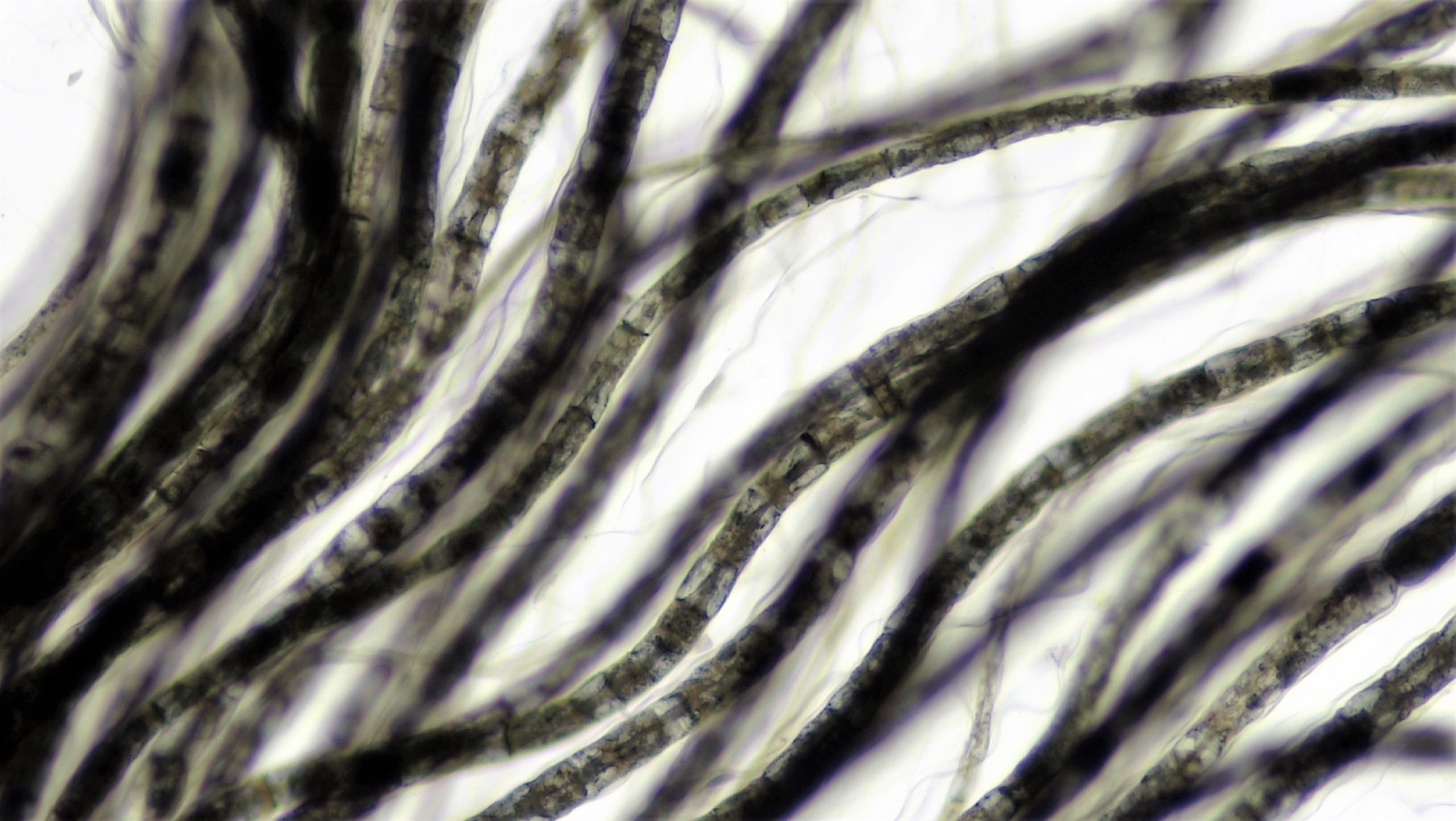
Featured above: Since osmium is attracted to oils in human tissue, it can be used to stain cells and make them more visible under a microscope. That’s precisely what’s going on with this bundle of nerves.
Show Notes
Once again I’m late to the blog post!! Here is a link to the promised explanation of relative proportion vs abundance — citations and further notes will have to wait a bit.
I have added all of my sources, but I’m afraid I haven’t had time to come back and write the show notes in a way I would have wanted to. However, I still wanted to add this link to BuzzFeed’s guidelines on writing about mental health, because they’re some of the best in the business.
And, in that spirit, the National Suicide Prevention Lifeline is 1-800-273-8255. Other international suicide helplines can be found at befrienders.org.
Episode Script
Osmium is a metal that knows how to make itself scarce. In the 1980s, NASA wanted to conduct spectroscopy in the extreme ultraviolet range using instruments on board the space shuttle. Element 76 is one of the few materials that’s capable of reflecting light in this highly energetic range — more than twice as good as gold or platinum. So the folks in charge of the mission sent up mirrors with a coating of pure osmium… and when they came back down, the osmium was gone.1 2
Like a lot of metals, pure osmium readily oxidizes when it comes in contact with the atmosphere. When it does, the most common product is OsO4, osmium tetroxide, a substance that is very interesting, and highly volatile. In other words, it easily transitions from a solid to a vapor. Really easily. Even hundreds of kilometers above the Earth, there was enough oxygen to convert the osmium to the oxide, which drifted away on the whisper-thin breeze.3
It’s not much easier to find back on the ground, either. Depending on how you measure it, osmium qualifies as the single rarest element on Earth — at least among the non-radioactive elements. (Many of those don’t exist anywhere in the natural universe at all — as far as we can tell.)4 5 6
That makes life a little difficult for an element hunter, but maybe that’s all for the best. If osmium were more plentiful on Earth, most people would probably go pretty far out of their way to avoid it.
You’re listening to The Episodic Table Of Elements, and I’m T. R. Appleton. Each episode, we take a look at the fascinating true stories behind one element on the periodic table.
Today, we’re getting our grubby hands all over osmium.
Does the name Smithson Tennant ring a bell? It’s been a while since we’ve glanced in his direction. He and his friend William Hyde Wollaston ran a clandestine international platinum smuggling ring and refinery out of Wollaston’s at-home secret laboratory. Their venture was so successful that for about twenty years, they retained exclusive control over almost every milligram of platinum that reached English shores — and no one found out about it until his death.
But we heard about all that thirty episodes ago. Besides, that’s Wollaston’s story. Tennant fronted some of the money, but after they figured out the purification process, he was relatively uninvolved with the day-to-day operation.
He still kept pretty busy, though. One of the byproducts of their platinum work was a dark and insoluble residue. Conventional wisdom said that this was simply graphite — the same old carbon you can find in a pencil. Tennant thought there might be something more in there, though, so he grabbed Wollaston by the arm and made haste to the secret lab.7 8 9 10
They were spurred on by stiff competition from another twosome on the opposite side of the Channel: Antoine-Francois le comte de Fourcroy and Nicolas Louis Vauquelin. The French team succeeded in producing the oxide of a new element first, but the English gents were the first to isolate the pure element. They quickly realized that they scored a twofer: elements 76 and 77. We’ll stick a pin in the latter for now.11
There’s not a ton of color variance among the metals of the periodic table. There’s gold and copper, obviously, but beyond that, they tend to land somewhere on the spectrum from “dull grey” to “shiny silver.” So the sliiight tinge of blue displayed by osmium is enough to make it stand out.12 13
Holding a sample of osmium is enough to reveal a far more noticeable property: It’s incredibly heavy. It is, in fact, the densest metal of all — actually, the densest of all the elements, or any other substance, for that matter. Nothing can pack the atoms together quite as tightly as osmium.
Technically, though, it’s not the densest thing that’s out there. In space, when a supergiant star reaches the end of its life, it can turn into a neutron star: A collection of material that is crushed so tightly together that, at its very center, it no longer makes sense to use terms like “elements” and “atoms.” Astronomers instead resort to calling the stuff “degenerate matter” and “nuclear pasta.”
If you could somehow twirl a forkful of nuclear pasta here on earth, that small amount would weigh nine times as much as the Great Pyramid of Giza. Actually — sorry, I forgot to move the decimal point there… it would actually have the same weight as nine hundred Great Pyramids of Giza. Definitely avoid it if you’re trying to lose a little weight.
Osmium, meanwhile, has a density of about 22.6 kilograms per liter. That might sound piddly after all that stellar nonsense, but it’s twice as dense as lead. Since the density of water is one kilogram per liter, that means a one-liter bottle of osmium would weigh even more than one of those five-gallon jugs from an office water cooler.14
While we’re on the subject, I would be remiss in my duties as a science podcaster if I didn’t point out a bit of a quirk: Kilograms are a measure of mass, not weight. Without getting too lost in the weeds, mass is a measure of how much stuff you have. A kilogram of osmium on Earth is the same as a kilogram of osmium on the Moon, and both are the same as a kilogram of osmium floating in the inky black void of space. Weight, meanwhile, is a measure of the force imposed upon an object by gravity, and the standard unit of measure for that is called the newton.
Since gravity is effectively constant everywhere on Earth, and since most folks spend most of their time on Earth, we can casually express weight in terms of kilograms as shorthand without really causing a problem.
So osmium is one hefty boi, but neither its feel nor its appearance struck Tennant the most. Those senses were overpowered by another. As he wrote:
When the alkaline solution is first formed … a pungent and peculiar smell is immediately perceived. This smell, as I afterwards discovered, arises from the extrication of a very volatile metallic oxide; and, as this smell is one of its more distinguishing characteristics, I should on that account incline to call the metal osmium.15
It’s a straightforward etymology relating to the Greek word for “scent” or “odor.” The original word is relatively neutral, pertaining to good and bad smells. Contrast with bromine, which comes from the Greek word for “stench.”16
It was the volatile tetroxide that filled Tennant’s nostrils, the same that disappeared from the mirrors on the space shuttle. Unlike osmium’s lexical forebear, however, this smell is pretty decidedly a bad thing. Aside from being just generally repugnant — similar to diesel oil, according to one researcher — it’s also extremely poisonous. The molecule easily binds with oils in our body, irritating the skin, nose, and throat. It’ll also burn the eyes, but it won’t stop there: it also permanently stains the corneas, rendering a person blind. Even at concentrations too low to smell — roughly two parts per billion — it can cause a fatal buildup of fluid in the lungs or damage the kidneys.17 18 19
Smithson Tennant managed to avoid those perils. He died when a bridge collapsed while he was crossing it, so his fate was less like Gilbert Lewis’ or Henri Moissan’s, and more like James Tiberius Kirk’s.20
Still, it’s rather nasty stuff, isn’t it? There’s something depressingly predictable about a substance as hazardous as this: Somebody, somewhere, has considered using it as a weapon.21
The United Kingdom’s Government Communications Headquarters, or GCHQ, is a sprawling and opaque intelligence-gathering organization that eavesdrops on internet traffic and telephone calls worldwide, roughly akin to the NSA in the United States. For more on that subject, see episode 68, Erbium.
In 2004, the GCHQ declared that they had uncovered a terrorist conspiracy to use the chemical in a coordinated attack on London’s subway network, Gatway airport, and shopping districts — an “osmium treason and plot,” if you will. ABC News reported, “Eight British citizens of Pakistani descent were arrested and taken into custody when 700 police raided 24 locations in and around London.”2223 24 25 26
None of those unnamed suspects actually possessed any osmium tetroxide.27 28
But people do tend to get pretty jumpy at the specific mention of an obscure and dangerous chemical like OsO4. For instance, in 2015, Royal Canadian Mounted Police arrested Christopher Phillips, a biochemical weapons specialist with possible mental health problems who was trafficking a suspicious package of hazardous chemicals after boasting about his plan to use them against the police.
Or at least, that’s how he was described by media outlets from the CBC29 to Chemistry World30 to the less esteemed Daily Mail. Phillips is a veteran who worked with osmium tetroxide during his time in the US Navy, but “biochemical weapons expert” seems a bit of a stretch. Since that time, he earned degrees in law and medicine and mainly worked as an ophthalmologist.
The accusation against him was based on a private email he had written to a friend. That email is three pages long, so there’s a lot to unpack, including his suggestion to unleash osmium tetroxide upon unwanted intruders in the home, especially if they happen to be police, and he recommended keeping it in a box labelled, “OSMIUM TETROXIDE — A BILLIONAIRE’S WEAPON OF TERROR.”31 32
However, he’s also pretty explicit in mentioning that his supply of the chemical was a result of his professional work, and that his friend might appreciate having a sample of the unique substance– stored safely under thick borosilicate glass inside a stainless steel box. In explaining why he was doing any of this in the first place, he wrote,
“It is probably … best for me to come up with a way to SAFELY disperse of at least part of my stockpile of this little novelty chemical. … knowing my luck, it could be improperly misconstrued as actual evidence.” Regarding the email itself, he wrote, “Seeing as how this is a written document that could potentially be stored for perpetuity, I do wish to stress that the box will not be designed to be actually used as a weapon.” He also expressed appreciation for the axe his friend kept under glass in case of “zombie apocalypse.”
These days, the RCMP has more in common with the GCHQ than with Dudley Do-Right, but they didn’t discover Phillips’ correspondence thanks to some comprehensive surveillance apparatus. It came to their attention after Phillips’ estranged wife forwarded them a copy of the email.33
The package of which the police were suspicious didn’t actually include any hazardous chemicals, nor anything incriminating at all.34
And for what it’s worth, according to Johns Hopkins University, more than one out of every four Americans deals with a diagnosable mental disorder in a given year.35 A 2014 paper published in the American Journal Of Public Health showed that those diagnosed with a mental illness were more likely to be the victims of violent crime than perpetrators of it.37
The judge in Phillips’ case said that a reasonable person would not see menace in his email, and there was no other evidence to support the charges of uttering threats against the police and possessing a dangerous weapon. He was cleared of both charges — but only after spending five months in prison.38 39 40
The main point I’m trying to make is not about Christopher Phillips, nor the RCMP, nor the GCHQ. I mention these stories because, if I am going to recommend that you provide personally identifying information over the internet in exchange for a dangerous substance to add your eccentric collection, you should know that it involves certain risks that have nothing to do with chemistry.
Refrain from making any flippant comments during the transaction and you should be fine, but you can avoid the trouble altogether by acquiring osmium in one of its more inert forms. We have a few different options.
Because osmium tetroxide is attracted to the oils in human skin, it can be used to reveal fingerprints left behind at the scene of a crime. It hasn’t actually been used for this purpose in decades, though, since the same thing can be achieved by safer means.41 42
Like other metals in the platinum group, osmium has been used for both the nibs of fountain pens and the balls of ball-point pens, both of which need to be durable enough to be pressed against paper for miles at a time. Often this takes the form of an alloy with the next element on the periodic table, which has the satisfyingly smooth name Osmiridium.
Lastly, there was a brief period of time when osmium was used for the tips of phonograph needles for the same reason it’s used in pens. That was basically just a fad, though. For a long time, record needles have been tipped with either diamond or sapphire. Outside of the jewelry business, the small amounts needed for this application are not very expensive at all.
If you choose to go this route, you’re practically guaranteed to be purchasing an artifact from a prior era with a distinct visual style. It would look quite fitting on a shelf next to that box of rhenium flashbulbs and that old tin can.43
Of course, for the discerning collector, only a sample of the pure thing will do. It’s not difficult to find such a thing. Arc-melted beads of osmium pop up pretty regularly online. If you acquire one of these, or something similar, you’ll need to store it carefully — inside a sealed glass container, preferably with secondary containment on top of that. You’ll also want to keep your sample in an inert environment, like mineral oil or argon gas. If you don’t, your osmium could eventually sublimate away, leaving you with naught but an invisible cloud of osmium tetroxide.44
After everything we’ve learned in this episode, I’m sure we can agree: that would probably be a bad idea.
Thanks for listening to The Episodic Table of Elements. Music is by Kai Engel. To learn the difference between relative portion versus abundance visit episodic table dot com slash O s.
Next time, we’ll send up a flare for iridium.
Until then, this is T. R. Appleton, reminding you that it might not be fair, but the internet is forever.
Sources
- Applied Optics, Low Earth Orbit Environmental Effects On Osmium And Related Optical-Film Coatings. T. R. Gull, H. Herzig, J. F. Osantowski, and A. R. Toft, August 1985.
- Applied Optics, Osmium Coated Diffraction Grating In The Space Shuttle Environment – Performance. M. R. Torr, September 1985.
- NASA’s Ask The Astronomer, Where Does The Atmosphere End And Outer Space Begin? Sten Odenwald, 1997.
- PeriodicTable.com, Abundance In Earth’s Crust Of The Elements. Theodore Gray.
- Cambridge Crystallographic Data Centre, Osmium.
- ScienceDirect, Osmium.
- Prabook, Smithson Tennant.
- Encyclopedia Britannica, Osmium. Last updated December 18, 2019.
- Encyclopedia.com, Tennant, Smithson.
- Mental Itch, The Interesting Discovery Of Osmium.
- Platinum Metals Review, Nicolas Louis Vauquelin: Early Work On Iridium And Osmium. W. A. Smeaton, 1963.
- PeriodicTable.com, Color Of The Elements. Theodore Gray.
- Encyclopedia.com, Osmium. Last updated February 5, 2021.
- Periodic-Table.org, Osmium — Density — Os. Nick Connor, November 21, 2020.
- Elementymology And Elements Multidict, Osmium. Peter van der Krogt.
- Etymonline, Bromine.
- New Jersey Department Of Health And Senior Services, Hazardous Substance Fact Sheet: Osmium Tetroxide. Last revised August 2002.
- LSU Health New Orleans, SOP For The Safe Use Of Osmium Tetroxide.
- UCLA Chemistry & Biochemistry, Standard Operating Procedure Working With Osmium Tetroxide.
- I can’t find who originally said it, but, “It’s ironic that after all those years of the crew falling down on the bridge, Captain Kirk died after a bridge fell on him.”
- The Israel Medical Association Journal, Osmium Tetroxide: A New Kind Of Weapon. Igor Makarovsky et. al., November 2007.
- ABC News, Bomb Plot Involved Deadly Chemical. Brian Ross and Christopher Isham, January 7, 2006.
- The Independent, Terror Gas Attack On Tube Foiled By Security Agencies. Kim Sengupta, October 10, 2011.
- The Guardian, Chemical Bomb Plot Uncovered. Richard Norton-Taylor and Rosie Cowan, April 6, 2004.
- BBC News, Chemical ‘Bomb Plot’ In UK Foiled. April 6, 2004.
- CNN.com, Sources: UK Chemical Attack Foiled. April 7, 2004.
- Globalization And WMD Proliferation: Terrorism, Transnational Networks And International Security, this page Edited by James J. Wirtz and James A. Russell, 2009.
- Unpeople: Britain’s Secret Human Rights Abuses, p. 104. Mark Curtis, 2008.
- CBC, Christopher Phillips’s Past Told Through U.S. Court Documents. Susan Allen and Melissa Mancini, January 23, 2015.
- Chemistry World, Ex-US Navy Weapons Specialist Arrested Over Chemical Stockpile. Rebecca Trager, January 27, 2015.
- Email written by Christopher Phillips, December, 2014.
- CBC News, Christopher Phillips Email Describes Uses For Osmium Tetroxide. Blair Rhodes, April 27, 2015.
- Yahoo! News, Christopher Phillips Email Describes Uses For Osmium Tetroxide. April 27, 2015.
- CBC, Christopher Phillips Arrest: No Hazardous Materials Found At Ottawa Hotel. January 21, 2015.
- Johns Hopkins Health, Mental Health Disorder Statistics.
- Correlating mental illness with terrorism and murder is not only inaccurate, but injurious to everyone’s well-being.36The American Journal Of Public Health, Community Violence Perpetration And Victimization Among Adults With Mental Illnesses. Sarah L. Desmarais et. al., November 12, 2014. (Strangely, this paper was accepted by the journal over a year earlier, on September 11, 2013. I’m not accustomed to seeing such a delay.)
- KSL.com, US Man In Canadian Chemical Case Found Not Guilty. The Associated Press, June 24, 2015.
- Toronto Star, Judge Finds Man Not Guilty Of Chemicals Threat, Pointing To Lack Of Evidence. Michael Tutton, June 24, 2015.
- CBC, Christopher Phillips Found Not Guilty Of Chemical Scare Charges. June 24, 2015.
- Journal Of Criminal Law And Criminology, The Use Of Hydrogen Fluoride In The Development Of Latent Fingerprints Found On Glass Surfaces. Herbert L. MacDonell, Winter 1960.
- Fingerprints And Other Skin Ridge Impressions, p. 151. Christophe Champod, Chris J. Lennard, Pierre Margot, and Milutin Stoilovic, 2004.
- See?
- The University Of Melbourne Scientific Scribbles, Osmium (Os): A Noble Metal In Character, But With Beastly Properties And A Heart Of Gold. Robin Sanchez Arit, August 11, 2017.
Such a fun podcast. Thank you, TR Appleton.
Thank you so much for listening — and for taking the time to comment! 🙂